In a groundbreaking fusion of biology and nanotechnology, scientists have harnessed the natural assembly capabilities of bacteriophages—viruses that infect bacteria—to create highly sensitive quantum dot-based sensors. Dubbed "nano-welders," these engineered phages act as molecular scaffolds, precisely positioning quantum dots to form functional devices with applications ranging from medical diagnostics to environmental monitoring. This innovative approach leverages billions of years of viral evolution to solve one of nanotech's most persistent challenges: the reliable, large-scale assembly of nanomaterials.
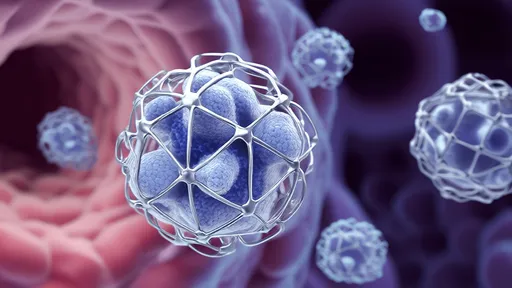
The key breakthrough lies in modifying the M13 bacteriophage, a filamentous virus whose coat proteins naturally bind to specific inorganic materials. By genetically programming these proteins to attract quantum dots—semiconductor nanoparticles with exceptional optical properties—researchers have created self-assembling hybrid structures. What makes this system extraordinary is its dual biological and electronic functionality. The phage's structural precision ensures quantum dots align at optimal distances for energy transfer, while its biological components enable selective binding to target molecules.
At the University of California, Berkeley, Dr. Elaine Chen's team demonstrated how these phage-quantum dot hybrids can detect trace heavy metals in water. "The viruses act like molecular breadcrumbs," Chen explains. "They not only organize the quantum dots into perfect arrays but also guide them to specific contaminants. When lead or mercury binds to the phage's surface receptors, it triggers measurable changes in the quantum dots' fluorescence—we're essentially hijacking viral mechanics for environmental sensing."
Medical applications appear equally promising. Harvard's Wyss Institute recently adapted the technology to identify cancer biomarkers in blood samples. Their design incorporates two quantum dot colors: one bound to phages targeting tumor proteins, another serving as an internal reference. The resulting color ratio provides quantitative measurements at sensitivities surpassing conventional ELISA tests. Unlike synthetic nanoparticle assemblies, these viral constructs remain stable in biological fluids—a critical advantage for clinical use.
The manufacturing potential could revolutionize nanoelectronics. Traditional lithography struggles to position quantum dots with the precision achieved by phage self-assembly. MIT researchers have already created prototype memristors using phage-aligned quantum dots, showing switching speeds 100x faster than graphene-based versions. "Nature's had a 3-billion-year head start in nanotechnology," remarks materials scientist Dr. Rajiv Singh. "We're just beginning to understand how to collaborate with biological systems rather than compete with them."
Challenges remain in scaling up production and ensuring batch consistency, but the field is advancing rapidly. Last month, a Korean team reported successfully "tuning" phage quantum dot spacing by altering viral DNA sequences—an essential step toward customized nanoarchitectures. Meanwhile, startup NanoVir Sensors has secured $20 million in funding to develop handheld detectors for foodborne pathogens using the technology.
Ethical discussions accompany these technical strides. Some biosecurity experts caution about potential dual-use risks, as the same principles could theoretically assemble harmful nanomaterials. However, proponents emphasize the technology's inherent safety: the phages used cannot infect human cells, and their quantum dot payloads are already FDA-approved for imaging applications.
Looking ahead, researchers envision expanding the phage "toolkit" to assemble diverse nanomaterials beyond quantum dots—from carbon nanotubes to superconducting particles. As synthetic biology converges with materials science, this viral assembly approach may well define the next generation of ultrasensitive, scalable nanodevices. The humble bacteriophage, long studied for its disease-causing abilities, has emerged as an unexpected ally in humanity's quest to master matter at the atomic scale.

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025