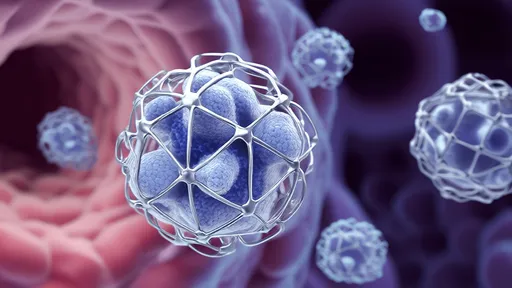
In a groundbreaking advancement for precision medicine, researchers have developed engineered exosomes capable of delivering therapeutic cargo directly to malfunctioning mitochondria. These tiny biological "couriers" – measuring just 30-150 nanometers – could revolutionize treatment for hundreds of mitochondrial disorders affecting 1 in 5,000 people worldwide.
The mitochondrial delivery challenge has long vexed scientists. These double-membraned organelles, often called cellular powerplants, are notoriously difficult to target. "It's like trying to mail a package to a specific room inside a locked building," explains Dr. Elena Vasquez from MIT's Synthetic Biology Center. "The outer cell membrane is the first barrier, then you need to escape endosomes, and finally penetrate the mitochondrial membranes."
Traditional approaches using viral vectors or lipid nanoparticles often fail to reach mitochondria intact. This new technique harnesses the body's own extracellular vesicles – specifically exosomes derived from mesenchymal stem cells. The researchers engineered these natural carriers with three key modifications: mitochondrial-targeting signal peptides, enhanced membrane fusion proteins, and pH-sensitive "escape" domains.
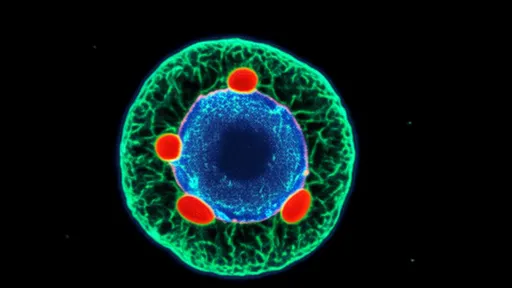
How the engineered exosomes work: The team decorated exosome surfaces with mitochondria-homing peptides derived from natural mitochondrial proteins. Once inside target cells, the pH-sensitive domains respond to acidic endosomal environments, triggering membrane fusion and cargo release. The mitochondrial targeting sequences then guide the payload through the organelle's complex double membrane.
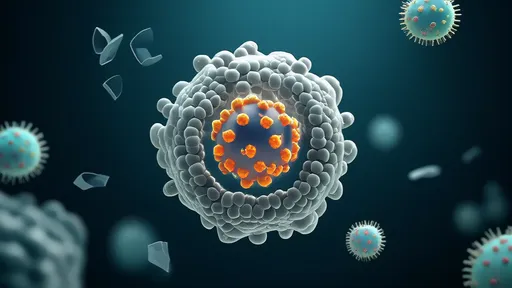
In proof-of-concept studies published in Nature Nanotechnology, the exosomes successfully delivered CRISPR-Cas9 components to mutate mitochondrial DNA in mice with inherited Leigh syndrome. Treated animals showed 60% improvement in motor function and 40% increase in ATP production compared to controls. "The precision is remarkable," notes Dr. Rajiv Patel from Cambridge University, uninvolved with the study. "They're achieving organelle-specific delivery with minimal off-target effects."
Beyond genetic editing, the platform shows promise for delivering other therapeutic payloads: antioxidant enzymes to combat oxidative stress, metabolic substrates for energy production, even engineered mitochondrial DNA itself. Early-stage trials are exploring applications in Parkinson's disease, where mitochondrial dysfunction plays a key role.
The technology also addresses a major hurdle in exosome therapeutics: consistent manufacturing. By developing a scalable bioreactor system that produces standardized exosome batches, the team has overcome previous limitations in yield and purity. "We can now generate clinical-grade exosomes with precisely controlled characteristics," says lead investigator Dr. Hiroshi Tanaka.
Safety considerations remain paramount when working with mitochondrial manipulation. The researchers incorporated multiple fail-safes: self-destructing payloads that degrade if mislocalized, dosage-dependent activation thresholds, and tissue-specific targeting ligands to prevent widespread distribution. Initial toxicology screens in primates showed no adverse effects at therapeutic doses.
Commercialization efforts are already underway, with startup MitoExo Therapeutics securing $53 million in Series B funding. The company plans to file IND applications for their lead candidate (targeting MELAS syndrome) by late 2024. Meanwhile, academic labs are exploring even more sophisticated designs – some incorporating AI-predicted targeting motifs or stimulus-responsive release mechanisms.
As the field progresses, regulatory frameworks struggle to keep pace. The FDA recently convened a working group specifically addressing exosome-based mitochondrial therapies. "This blurs traditional categories between gene therapy, drug delivery, and medical devices," acknowledges FDA Commissioner Dr. Robert Califf during a recent symposium.
The implications extend far beyond rare genetic disorders. Age-related mitochondrial decline contributes to nearly all chronic diseases of aging. If delivery efficiency can be further improved, this technology might eventually help maintain cellular energy production in aging populations. Some researchers even speculate about performance enhancement applications – though ethical guidelines currently prohibit such use.
While challenges remain in scaling production and demonstrating long-term efficacy, the scientific community largely agrees this represents a paradigm shift. "We're no longer just treating symptoms of mitochondrial disease," reflects Dr. Vasquez. "For the first time, we can precisely target the root cause inside these vital organelles." As clinical trials progress, this mitochondrial "FedEx" system may soon deliver hope to patients with previously untreatable conditions.

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025