In the relentless battle against cancer metastasis, scientists have turned to nature for inspiration, developing a groundbreaking "dormancy cage" strategy using bioinspired hydrogels. This innovative approach aims to physically contain disseminated tumor cells within a synthetic extracellular matrix, effectively putting metastatic seeds into a state of suspended animation. The technology represents a paradigm shift from traditional cytotoxic approaches to metastasis prevention, focusing instead on creating artificial niches that mimic the body's natural tumor-suppressive microenvironments.
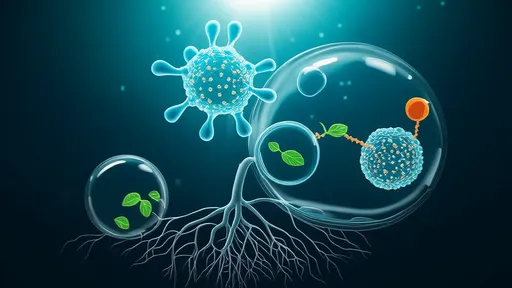
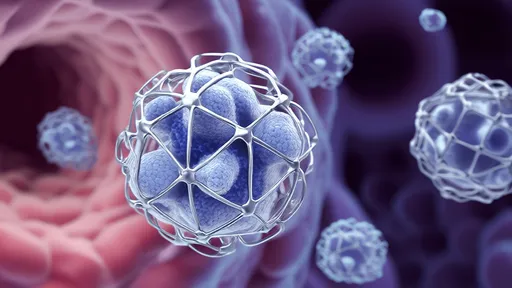
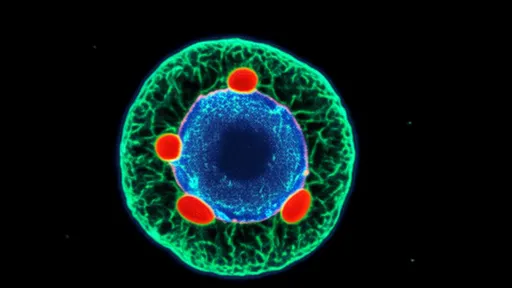
The hydrogel's secret lies in its remarkable biomimicry – its structure replicates the mechanical and biochemical properties of bone marrow stroma, where dormant disseminated tumor cells naturally reside. By engineering precise viscoelasticity and incorporating dormancy-inducing signaling molecules, researchers have created what they describe as a "physical and biological prison" for cancer cells. Early-stage tumor cells captured within this matrix enter prolonged quiescence, unable to proliferate or escape, while remaining vulnerable to future targeted therapies.
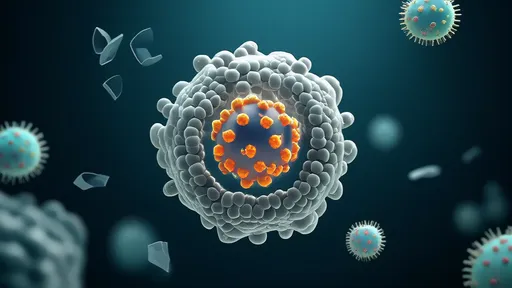
What sets this approach apart is its dual-action mechanism. Unlike conventional treatments that attack cancer cells directly, the hydrogel acts as a biological decoy, tricking tumor cells into believing they've reached a protective niche where aggressive growth would be disadvantageous. Simultaneously, the material's nanostructured fibers form a physical barrier that prevents circulating tumor cells from extravasating into vulnerable tissues. This combination of biological signaling and mechanical confinement creates what researchers poetically call a "gilded cage" for metastatic cells.
The technology's development required solving numerous challenges in materials science and tumor biology. Scientists had to precisely tune the hydrogel's pore size to allow nutrient diffusion while preventing cell migration, a balance comparable to creating a sieve that distinguishes between water molecules and microscopic invaders. Additionally, the team incorporated enzyme-responsive components that strengthen the matrix when exposed to tumor-associated proteases, creating a self-reinforcing barrier against increasingly aggressive cancer cells.
Clinical implications could be profound if human trials confirm animal study results. Patients with surgically removed primary tumors might receive hydrogel implants at potential metastatic sites, creating protective zones that intercept wandering cancer cells. The approach could buy critical time for adjuvant therapies to work and potentially prevent metastasis altogether. Researchers emphasize this isn't a cure, but rather a game-changing metastasis management strategy that could transform many cancers into chronic but controllable conditions.
Ongoing research focuses on optimizing the hydrogel's residence time and developing non-invasive methods to monitor contained tumor cells. Some versions now include contrast agents for MRI tracking and thermally responsive components that allow localized drug release when activated by external energy sources. These smart hydrogel systems represent the next frontier in metastasis prevention – materials that don't just passively contain cancer cells, but actively communicate with clinicians about the status of their microscopic prisoners.
The scientific community has greeted these developments with cautious optimism. While acknowledging the significant hurdles remaining before clinical deployment, experts agree the approach offers a fundamentally new weapon against metastasis – cancer's most lethal capability. By learning from how the body naturally controls wayward cells rather than relying solely on pharmacological assault, researchers may have found a way to turn cancer's own survival strategies against itself, trapping metastatic seeds in engineered sanctuaries where they can do no harm.

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025