In a groundbreaking development at the intersection of biotechnology and wound care, researchers have unveiled a novel "smart bandage" powered by fungal mycelium. This living material, derived from the intricate root-like networks of fungi, demonstrates remarkable potential in accelerating tissue regeneration while actively preventing infections. The innovation emerges from years of studying how certain fungi interact with biological systems, revealing properties that could revolutionize chronic wound treatment.
The mycelium-based dressing represents a significant departure from traditional passive wound coverings. Unlike conventional bandages that merely protect injuries from external contaminants, this fungal-derived matrix actively participates in the healing process. Laboratory tests show the material stimulates fibroblast activity and collagen production while maintaining optimal moisture levels – a critical factor often overlooked in standard wound care protocols. Early-stage clinical observations suggest healing times may be reduced by as much as 40% for certain types of ulcers and surgical wounds.
What makes this biological bandage particularly innovative is its self-regulating antimicrobial properties. The mycelium network naturally secretes compounds that inhibit bacterial growth without relying on pharmaceutical antibiotics. This characteristic addresses the growing concern of antibiotic resistance in chronic wound infections. Researchers have identified several bioactive molecules within the fungal material that appear to selectively target pathogenic bacteria while preserving beneficial microorganisms essential for proper skin recovery.
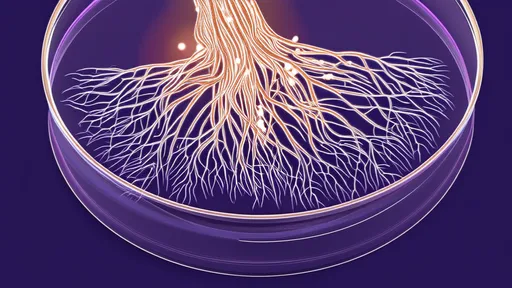
The production process involves cultivating specific fungal strains under controlled conditions to create a dense, leather-like mat. Through proprietary techniques, scientists can tune the material's porosity, thickness, and degradation rate to match different wound types. As the wound heals, the fungal material gradually breaks down into biocompatible compounds that the body safely absorbs, eliminating the need for painful dressing changes that can disrupt delicate new tissue growth.
Perhaps most astonishing is the material's responsive behavior to wound conditions. The living components within the bandage appear to modulate their metabolic activity based on pH changes and biochemical markers present in the wound bed. During periods of increased inflammation, the fungal network releases higher concentrations of anti-inflammatory compounds. This dynamic interaction creates what researchers describe as a "dialogue" between the dressing and the wound environment – a level of sophistication unmatched by synthetic wound care products.
Current research focuses on optimizing strain selection and growth parameters to enhance the material's therapeutic effects. Particular attention is being paid to fungal species that produce compounds known to stimulate angiogenesis – the formation of new blood vessels crucial for delivering oxygen and nutrients to healing tissues. Early prototypes have shown promise in treating diabetic foot ulcers, a condition that leads to approximately 70,000 lower-limb amputations annually in the United States alone.
The development team emphasizes that while the technology appears futuristic, it actually builds upon ancient relationships between fungi and other organisms. Many of the observed healing mechanisms mirror natural processes that occur when certain fungi form symbiotic relationships with plant roots in soil ecosystems. This biomimetic approach lends credibility to the technology's safety profile and suggests we may be rediscovering biological partnerships that evolution perfected over millennia.
Regulatory pathways for this novel medical device present unique challenges, as it represents a hybrid between a living organism and a traditional wound dressing. Researchers are working closely with health authorities to establish appropriate classification frameworks and safety standards. Small-scale human trials could begin within two years, pending further successful animal studies and manufacturing scale-up.
Beyond acute wound care, scientists speculate about potential applications in burn treatment, post-operative recovery, and even cosmetic dermatology. The material's ability to support three-dimensional tissue growth has prompted investigations into its use as scaffolding for skin grafts and engineered tissue constructs. Some researchers envision future versions incorporating genetically modified fungal strains designed to deliver targeted drug therapies or growth factors based on real-time wound diagnostics.
As climate change increases concerns about sustainable medical technologies, the fungal bandage offers environmental advantages. Production requires minimal energy compared to synthetic alternatives, and the material is fully biodegradable. This aligns with growing demands for healthcare solutions that reduce medical waste without compromising patient outcomes. The technology's scalability could make it particularly valuable in resource-limited settings where advanced wound care remains inaccessible.
The scientific community has greeted these developments with cautious optimism. While acknowledging the need for rigorous clinical validation, experts agree that mycelium-based therapies could represent a paradigm shift in wound management. The project's lead researcher summarizes the technology's promise: "We're not just covering wounds anymore – we're creating an active healing environment that works with the body's natural processes. This isn't science fiction; it's the future of regenerative medicine growing right before our eyes."

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025